Le concept de communication bidirectionnelle entre les organes de la cavité abdominale et le cerveau remonte à l'ère hippocratique. Ce n'est cependant qu'au cours du siècle dernier que les mécanismes neuro-endocriniens qui sous-tendent cette connexion ont été décrits et plus récemment encore, leur pertinence pour la physiopathologie de la neurodégénérescence a été appréciée.
Braak et d'autres ont fait l'hypothèse que cette communication se fait par l'intermédiaire du nerf vagal, d'une certaine façon tous les nerfs sont des conduits pouvant véhiculer des microbes, prions et virus. Cet article fait l'hypothèse que l'axe intestin-cerveau influence la production d'acides biliaires par le microbiome intestinal.
 Le catabolisme du cholestérol est la source des acides biliaires.
Le catabolisme du cholestérol est la source des acides biliaires.
Le cerveau est l'organe le plus riche en cholestérol car il contient environ un quart du cholestérol total présent dans le corps. Le cholestérol et les molécules lipidiques apparentées sont un constituant crucial des neurones et des celluls nerveuses non neuronales. Il est aussi un composant majeur de la myéline. La myéline est un matériau riche en lipides qui entoure les axones des cellules nerveuses. Étonnamment, les acides biliaires peuvent également être produits dans le cerveau, où ils régulent le métabolisme cellulaire des lipides et du glucose.
Le cerveau contient de nombreux acides biliaires conjugués et non conjugués qui pénètrent dans le cerveau soit par diffusion, soit par transport actif via des transporteurs d'acides biliaires à partir de la circulation systémique, soit par synthèse locale dans les astrocytes et les neurones .
Pratiquement tout le cholestérol cérébral est formé localement, cependant, dans le cerveau adulte, la synthèse du cholestérol est faible en raison d'un recyclage efficace. La demi-vie du cholestérol cérébral est estimée à au moins 5 ans.
La fonction hépatique se détériore cependant avec l'âge et, par conséquent, l'homéostasie des acides biliaires change, avec une synthèse réduite des acides biliaires et une augmentation de l'accumulation de cholestérol périphérique (Bertolotti et al., 2007). Cependant, le métabolisme du cholestérol est différent dans le cerveau par rapport aux organes périphériques (Smiljanic et al., 2013).
Jusqu'à il y a quelques décennies, on pensait que les acides biliaires servaient uniquement de surfactants pour aider le corps à absorber les graisses, les vitamines liposolubles et les stéroïdes dans la circulation sanguine afin que le foie puisse les traiter.
Cependant, il est maintenant entendu que les acides biliaires, en plus d'agir comme agents émulsifiants, ont une biologie complexe qui affecte une variété de processus physiologiques et qu'ils sont des molécules de signalisation de type hormonal (Zangerolamo, Vettorazzi, Rosa, Carneiro et Barbosa, 2021 ).
Les niveaux d'acides biliaires doivent être étroitement surveillés car ils peuvent devenir toxiques. Il est essentiel de comprendre le rôle des acides biliaires dans la physiopathologie des maladies neurodégénératives dont la maladie de Parkinson, la maladie d'Alzheimer, la SLA (maladie de Lou Gherig/Charcot) et autres.
Acides biliaires primaires
La première étape de la synthèse des acides biliaires est la génération d'acides biliaires primaires, dont la majorité se produit dans le foie pour finalement former des sels biliaires primaires conjugués d'acide cholique (CA) et d'acide chénodésoxycholique (CDCA).
Acides biliaires secondaires
Les acides biliaires secondaires sont formés par des modifications enzymatiques des acides biliaires primaires par des bactéries présentes dans le côlon où ils servent de substrats pour le métabolisme microbien. Le microbiome intestinal influence les composants du pool d'acides biliaires et les acides biliaires peuvent réguler la composition des bactéries intestinales par des effets antimicrobiens directs et indirects.
La déconjugaison microbienne des acides biliaires primaires conjugués à la taurine et à la glycine via l'hydrolase des sels biliaires est une fonction hautement conservée dans les phylae et les archées bactériennes et aide à diminuer l'effet microcide des acides biliaires intraluminaux. Les bactéries avec des pompes d'exportation de sels biliaires ont un avantage sélectif supplémentaire dans cet environnement.
Malgré leurs caractéristiques qui en font des troubles distincts, de nombreuses maladies neurologiques partagent des caractéristiques pathologiques communes telles que des dépôts de protéines mal repliées et une neuroinflammation en plus de la perte neuronale et de la fonction cellulaire perturbée comme le dysfonctionnement mitochondrial et le stress oxydatif.
Récemment, les acides biliaires et leurs récepteurs ont été associés à des processus pathologiques dans de nombreuses maladies neurologiques et les acides biliaires se sont révélés prometteurs en tant que traitements putatifs pour un certain nombre de ces troubles.
Le vieillissement
Le vieillissement, qui est le plus grand risque de troubles neurodégénératifs, peut également entraîner plusieurs changements dans l'intestin qui peuvent avoir un impact sur l'axe microbiome intestinal-cerveau, notamment une stabilité et une diversité réduites des communautés microbiennes, des niveaux accrus d'inflammation et d'amincissement de la muqueuse et une réduction biodisponibilité des métabolites microbiens avec des actions immunorégulatrices telles que les acides biliaires secondaires et les acides gras à chaîne courte.
La maladie de Parkinson
Un certain nombre d'études précliniques chez les rongeurs ont fourni des preuves que les acides biliaires pourraient être impliqués dans la pathogenèse de la maladie de Parkinson.
Une analyse du métabolome du cerveau de souris recevant des injections intracérébrales de fibrilles d'α-synucléine préformées dans le bulbe olfactif a montré des altérations des voies du métabolisme de la taurine qui pourraient affecter la conjugaison taurine de l'acide tauroursodésoxycholique (TUDCA) et de l'acide ursodésoxycholique (UDCA) (Graham et al., 2018 ; Graham et al., 2018).
Les acides biliaires primaires (CA, MCA total et β-MCA) et les acides biliaires secondaires (TUDCA, acide taurohyodésoxycholique (THCDA) et DCA) ont été augmentés dans les échantillons fécaux. Le traitement de rats atteints de la maladie de Parkinson induite par la roténone avec de l'UDCA a restauré les niveaux de dopamine striatale et normalisé les marqueurs d'inflammation et d'apoptose.
Deux essais cliniques sont actuellement en cours (NCT02967250 et NCT03840005) pour évaluer l'efficacité de l'UDCA comme traitement pour les patients atteints de la maladie de Parkinson.
Cependant, le rôle du taux de cholestérol dans la pathogenèse de la maladie de Parkinson est loin d'être clair. La plupart des études publiées ont révélé qu'un taux de cholestérol élevé était associé à une prévalence réduite de la maladie de Parkinson et que le traitement aux statines pour réduire le cholestérol augmentait le risque de maladie de Parkinson (Liu et al., 2017).
Cela va à l'encontre de l'opinion dominante selon laquelle les statines sont des agents neuroprotecteurs et des études récentes chez des patients atteints de la maladie de Parkinson ont produit des résultats contradictoires.
La raison des résultats différents reste inconnue, mais pourrait être liée à une hydrophobicité différente des statines, les statines hydrophiles mais non lipophiles étant associées à une progression plus rapide de la maladie de Parkinson (Lewis et al., 2022). Un essai récent (NCT02787590) de la simvastatine pour ralentir la progression de la maladie de Parkinson n'a montré aucun avantage (https://cureparkinsons.org.uk/2020/09/simvastatin-results/).
La maladie d'Alzheimer
Des rapports modifiés de 23 acides biliaires sériques ont été associés à des biomarqueurs du liquide céphalo-rachidien de la maladie d'Alzheimer (Nho et al., 2019). De plus, les taux plasmatiques d'ACL (acide lithocholique) ont augmenté chez les patients atteints de la maladie d'Alzheimer par rapport aux témoins et les taux plasmatiques d'acide glycochénodésoxycholique (GCDCA), d'acide glycodésoxycholique (GDCA) et d'acide glycolithocholique (GLCA) étaient significativement élevés chez les patients atteints de la maladie d'Alzheimer par rapport aux patients atteints de troubles cognitifs légers. malades en situation de handicap.
Baloni et ses collègues (Baloni, et al., 2020) ont signalé des augmentations similaires des acides biliaires dans les échantillons de cerveau post-mortem de la maladie d'Alzheimer, ainsi qu'un rapport accru de GCDCA: CA et une augmentation des acides biliaires secondaires DCA, LCA, TDCA et GDCA dans la maladie d'Alzheimer. échantillons.
Cela s'est accompagné de niveaux plus élevés de taurine sérique. Comme la synthèse secondaire des acides biliaires ne se produit pas dans le cerveau, cela suggère que les modifications du sérum et des tissus cérébraux peuvent refléter des altérations du microbiome intestinal des patients atteints de la maladie d'Alzheimer.
Des changements dans le métabolisme des androgènes ont été signalés chez des souris sans germes par rapport à des souris avec un microbiome intestinal normal où un intestin sain maintient des niveaux élevés de dihydrotestostérone (Collden et al., 2019). Des niveaux réduits de dihydrotestostérone sont signalés comme un facteur de risque de la maladie d'Alzheimer (Rosario & Pike, 2008) et peuvent réguler l'accumulation de β-amyloïde.
La sclérose latérale amyotrophique
On sait qu'AMX0035, un médicament récemment mis sur le marché dans certains pays, associe le sodium phenylbutyrate à l'ursodoxicoltaurine (taurursodiol).
Le cholestérol était élevé dans le liquide céphalo-rachidien des patients atteints de sclérose latérale amyotrophique et le (25R) 26-hydroxycholestérol (également connu sous le nom de 27-hydroxycholestérol) de la branche acide de la biosynthèse des acides biliaires a été réduit, ce qui a entraîné une incapacité du cerveau à éliminer l'excès de cholestérol (Abdel-Khalik et al., 2017). Cela pourrait indiquer une tentative de l'organisme d'atténuer la toxicité de l'excès de cholestérol par sa conversion en acides biliaires.
Les souris mutantes SOD1 présentent également une dysbiose du microbiote intestinal au stade pré-symptomatique (Figueroa-Romero et al., 2019).
De même, la plus grande étude à ce jour sur des patients humains atteints de sclérose latérale amyotrophique a révélé que la progression de la maladie était corrélée à une réduction de la diversité microbienne (Di Gioia et al., 2020). Dodge et ses collègues (Dodge et al., 2021) ont rapporté que le CDCA, le LCA, le TDCA et le TLCA (acide taurolithocholique) étaient élevés dans les fèces des souris SOD1 en phase terminale par rapport aux types sauvages. Chez les patients atteints de sclérose latérale amyotrophique, plusieurs genres de microbiote ont été signalés comme étant plus faibles, notamment des familles Lachnospiraceae et Ruminococcaceae (Martin, Battistini, & Sun, 2022).
Le métabolisme altéré du cholestérol dans la sclérose latérale amyotrophique peut avoir des effets à la fois sur les voies des acides biliaires dans le cerveau et l'intestin, bien que l'on ne sache pas encore lequel peut se produire en premier.
Pour contrer le stress mitochondrial et oxydatif impliqué dans la cause de la sclérose latérale amyotrophique, un essai clinique de phase II utilisant le traitement TUDAC avec le riluzole, un inhibiteur des récepteurs du glutamate, pendant 1 an a été mené pour évaluer l'efficacité du ciblage de la signalisation des acides biliaires comme stratégie thérapeutique. pour le traitement de la sclérose latérale amyotrophique et ont constaté que le traitement ralentissait la détérioration de la fonction et améliorait l'évaluation fonctionnelle de 15 % chez les patients atteints de la maladie (Elia et al., 2016).
Des études examinant les effets de TUDCA et d'autres acides biliaires sur la survie cellulaire et l'inversion des déficits dans des modèles animaux de sclérose latérale amyotrophique ont également montré des résultats encourageants.
Maladie de Huntington
Des preuves récentes ont suggéré qu'une dysbiose intestinale peut survenir chez les patients atteints de Huntington et certains genres de microbiote ont été corrélés à des scores cliniques spécifiques, à savoir Intestinimonas avec des scores de capacité fonctionnelle totale et Lactobacillus négativement corrélé avec des scores au mini-examen de l'état mental (Wasser et al., 2020 ).
Une dysbiose intestinale a été rapportée dans des modèles murins atteints de la maladie de Huntington avec une diversité α différentielle. Il est proposé qu'une diversité accrue puisse être associée à une volatilité accrue de la microglie intestinale et à une fonction perturbée du microbiome intestinal (Kong et al., 2021). Aucune étude n'a encore examiné les modifications des acides biliaires chez les patients atteints de la maladie de Huntington, mais étant donné les dysfonctionnements signalés dans la signalisation du cholestérol dans le cerveau et la dysbiose intestinale, il semble probable qu'ils se produisent et des modifications du pool d'acides biliaires pourraient altérer cette régulation. Pourtant, un essai clinique de phase I avec l'UDCA (NCT00514774) chez des patients atteints de la maladie de Huntington a débuté en 2007, mais les résultats de l'essai n'ont jamais été publiés.
Sclérose en plaques
Bhargava et ses collègues (Bhargava et al., 2020) ont mené une analyse métabolomique de patients atteints de différents types de sclérose en plaques et ont trouvé des niveaux inférieurs de métabolites primaires des acides biliaires chez les patients atteints de sclérose en plaques progressive primaire par rapport aux témoins sains et des niveaux inférieurs de métabolites secondaires des acides biliaires dans les groupes de sclérose en plaques progressive primaire et de sclérose en plaques récurrente-rémittente, indiquant des altérations du métabolisme des acides biliaires dans la sclérose en plaques qui étaient les plus importantes chez les patients atteints de sclérose en plaques progressive primaire.
Des résultats similaires ont été mesurés chez des patients atteints de sclérose en plaques pédiatrique par rapport à des témoins appariés selon l'âge, ce qui suggère que l'âge n'était pas un facteur dans l'altération du métabolisme des acides biliaires survenant dans la sclérose en plaques (Bhargava et al., 2020). Expérimentalement, les mêmes auteurs ont découvert que le traitement avec TUDCA et l'agoniste GPBA INT-577 prévenait la neuroinflammation (astrocytes réactifs et polarisation pro-inflammatoire de la microglie) de manière dose-dépendante dans les astrocytes et la microglie en culture. De plus, TUDCA a réduit les déficits comportementaux et les changements neuropathologiques chez les souris atteintes d'encéphalomyélite auto-immune expérimentale grâce à ses effets sur le GPBA.
Conclusion
Il est de plus en plus évident que la signalisation des acides biliaires est bidirectionnelle dans l'axe intestin-cerveau, signalant l'état métabolique et contribuant à l'homéostasie du cholestérol dans le cerveau.
De multiples cibles pharmacologiques ont déjà été développées pour une application dans les maladies du foie qui peuvent potentiellement être réutilisées pour une application à la neurodégénérescence. Ceux-ci peuvent agir de manière centrale dans le SNC ou exploiter les voies périphériques des acides biliaires pour affecter indirectement un changement.
Des travaux supplémentaires sont donc nécessaires pour déterminer la contribution relative de tous les acides biliaires et des espèces d'oxystérols dans la santé et la maladie neuronales afin d'éclairer les études interventionnelles et de développer une nouvelle catégorie de médicaments pour le traitement de la neurodégénérescence.
Read the original article
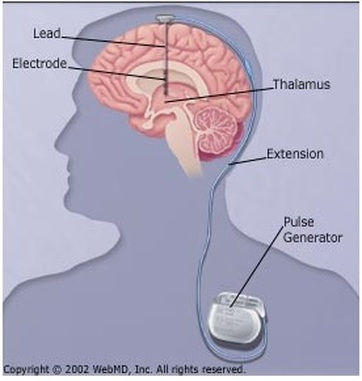 Deep brain stimulation of the subthalamic nucleus is a well-established treatment for the motor symptoms of Parkinson's disease, but it is still poorly understood how subthalamic stimulation modulates the state dynamics of the brain's motor network. A biological network represents systems as complex sets of relationships between various biological entities.
Deep brain stimulation of the subthalamic nucleus is a well-established treatment for the motor symptoms of Parkinson's disease, but it is still poorly understood how subthalamic stimulation modulates the state dynamics of the brain's motor network. A biological network represents systems as complex sets of relationships between various biological entities.
 The metabolic consequences of the decline in estrogen during menopause accelerate this neuropathology in women. However, the use of hormone replacement therapy gives contradictory results in the prevention of cognitive decline.
The metabolic consequences of the decline in estrogen during menopause accelerate this neuropathology in women. However, the use of hormone replacement therapy gives contradictory results in the prevention of cognitive decline.
 Source: Xocolatl via Wikipedia
Source: Xocolatl via Wikipedia Le catabolisme du cholestérol est la source des acides biliaires.
Le catabolisme du cholestérol est la source des acides biliaires.