Few people reach the age of 80 without a kind of neuropathology developing in their brains, and multi-pathology is the norm. Neuropathologists commonly observe cytoplasmic inclusions of phosphorylated TDP-43 in post-mortem brain samples with or without Aβ plaques and neurofibrillary tangles that define Alzheimer's disease.
Inclusions containing this RNA binding protein were first implicated in ALS and FTD more than 10 years ago. Shortly after, neuropathologists identified the TDP-43 pathology in people who had symptoms of Alzheimer's disease, not FTD (Amador-Ortiz et al., 2007). It often accompanied the hippocampal sclerosis, a disease characterized by a narrowed hippocampus ravaged by gliosis. More than a decade ago, Dickson had described curious cases of hippocampal sclerosis in people with dementia without the pathology of Alzheimer's disease (Dickson et al., 1994).
Researchers now suspect that many of these cases, as well as more recent cases, were caused by TDP-43 proteinopathy.
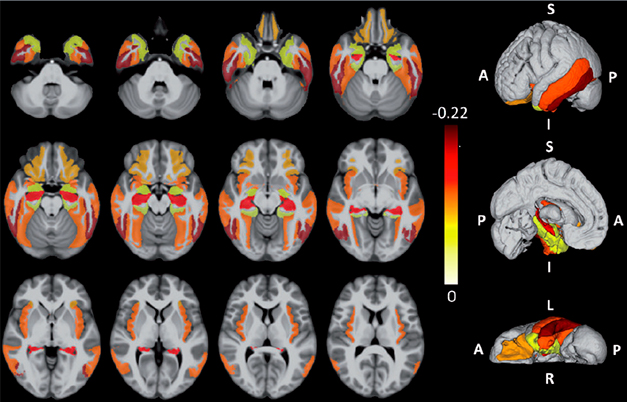
Over the last ten years, several studies have described the clinical and pathological evolution of this TDP-43 proteinopathy, recently called LATE. A study of TDP-43 in post-mortem brain samples of nearly 1,000 deceased participants in the Rush Memory and Aging Project and RUD-MAP (ROS-MAP) project (James et al. , 2016). Half of the participants had TDP-43 pathology and 37% had both TDP-43 and Alzheimer's disease. People with both conditions were more likely to have clinical dementia than those with only one condition. This study proposed a pathological staging pattern, in which the pathology TDP-43 reached the amygdala at stage 1, then the hippocampus and cortex entorhinal at stage 2, and finally the neocortex stage at 3. Once the pathology extends beyond the amygdala, it correlates well with cognitive impairment.
LATE is a proposal for a new common neurodegenerative disease, which has been named so by an international group of pathologists, clinicians and epidemiologists in a consensus report published April 30 in the Brain journal.
This international group predicts that this disease is a clinical diagnosis of Alzheimer's disease over five diagnosis. In their article, written after a workshop in Atlanta last fall, Peter Nelson's researchers at the University of Kentucky at Lexington summarized the decades of research that preceded the term. They describe the characteristics of LATE, propose ways to classify and diagnose it, and call for the development of specific biomarkers and therapies.
A typical case of predominantly age-related TDP-43 encephalopathy in limbic (LATE), would be that of an 86-year-old woman who would develop classic amnesic symptoms of Alzheimer's disease in her last years of life, but at autopsy, his brain would only carry a modest burden of Aβ plates and tau entanglement. Instead, the inclusions of TDP-43 would have invaded its limbic regions.
The LATE pathology often coincides with the hippocampal sclerosis, but the latter is not necessary for a diagnosis.
How often is LATE? ROSMAP researchers, led by Rush's Julie Schneider, analyzed their cohort data again to take into account the proposed neuropathological staging. They estimate the impact of LATE at about half that of Alzheimer's disease in the elderly and equal to the combined impact of all vascular neuropathologies. This would make LATE about 100 times more widespread than the FTD.
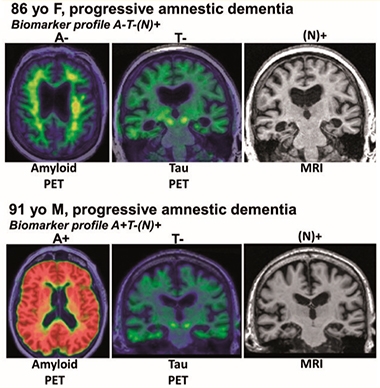
The co-occurrence of Alzheimer's disease and LATE neuropathologies in older people complicates the interpretation of therapeutic trials specific to Alzheimer's disease and may even mask positive results, Nelson noted.
The question is: what is the usefulness of introducing a new definite disease that can be diagnosed so far only through post-mortem neuropathological findings?
How will researchers differentiate the proposed new disease from Alzheimer's disease? There are no fluid biomarkers or PET tracers to detect the pathology of TDP-43, although researchers are interested. The intracellular localization of TDP-43 and its low pathological burden complicate the development of biomarkers. For the moment, the best the diagnoser can do is to go through a process of elimination.
Once biomarkers exist, they will facilitate targeted drug discovery efforts on LATE, for example on its TDP-43 aggregates. Given that comorbid neuropathologies in LATE at risk could potentially interact (Robinson et al., 2018), Trojanowski has proposed an immunotherapeutic cocktail of antibodies to Aβ, tau, TDP-43 and α-synuclein.

