The phase 2 LixiPark trial (NCT03439943) showed that treatment with lixisenatide (Adlyxin, Sanofi), a glucagon-like peptide-1 receptor agonist used as a therapy for diabetes, may have resulted in less progression of motor disability compared with placebo at 12 months in patients with early Parkinson disease (Parkinson’s disease) but was associated with gastrointestinal adverse effects.
 The results of this recent trial add further evidence to exenatide’s potential as a drug that might slow the progression of Parkinson’s and pave the way for the larger phase III clinical trial that is currently underway;
The results of this recent trial add further evidence to exenatide’s potential as a drug that might slow the progression of Parkinson’s and pave the way for the larger phase III clinical trial that is currently underway;
Diabetes mellitus is a risk factor for Parkinson's disease. UK's Cure Parkinson’s has been at the forefront of exenatide’s journey as a potential treatment for Parkinson’s from the outset. They funded the first clinical study of exenatide in people with Parkinson’s. This was a year-long pilot study in 2008 involving 45 people with Parkinson’s. Those who took exenatide did not experience the decline in their movement that is normally seen due to Parkinson’s. Actually, these participants improved a little. Crucially, some of these benefits were still present when measured one year after the participants had stopped taking exenatide, giving hope that this medicine had interfered with the underlying disease process, rather than simply masking symptoms.
Liraglutide is another drug that belongs to a class of medicines called Glucagon-like peptide 1 (GLP-1) receptor agonists. In addition to treating T2 diabetes and obesity, this class of drugs has another interesting property: taming inflammation. In animal models of Parkinson’s, liraglutide has shown strong neuroprotective effects.
Based on this, Cure Parkinson’s in 2017 funded Professor Michele Tagliati to undertake a phase II randomized, double-blind, placebo-controlled clinical trial of liraglutide in people with Parkinson’s.
70 participants were given either once-daily injections of liraglutide or placebo injections for 52 weeks. The study was primarily designed to assess changes in symptoms whilst off Parkinson’s medication for 12 hours before the assessments. Movement (motor), non-motor, and cognitive symptoms were assessed along with several secondary measures, including quality of life and daily activities. The primary analysis of the results included 37 people with Parkinson’s on liraglutide and 18 on the placebo drug.
Non-motor symptoms, activities of daily living, and quality of life appeared to significantly improve in the group on liraglutide treatment. However, there was no clear difference in motor symptoms between those on liraglutide and those on the placebo; it was noted that there appeared to be a strong placebo effect in this study, meaning the participants, even though none was aware they were taking the placebo drug, believed they were experiencing therapeutic results. This is consistent with the often-reported association of more invasive treatments causing a stronger placebo effect.
The research team also reported a significant lowering of body mass index (BMI) and average blood glucose levels in the active drug group, which are not desirable for patients in general. Interestingly, significant mobility improvements were reported by people taking liraglutide in their quality of life experiences, and this was more than in the placebo group.
In this new phase II of lixisenatide trial, the effect of lixisenatide was assessed on the progression of motor disability in persons with Parkinson’s disease.
Participants with Parkinson’s disease who were receiving a stable dose of medications to treat symptoms, and who did not have motor complications were randomly assigned in a 1:1 ratio to daily subcutaneous lixisenatide or placebo for 12 months, followed by a 2-month washout period. The primary endpoint was the change from baseline in scores on the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III (range, 0 to 132, with higher scores indicating greater motor disability), which was assessed in patients in the on-medication state at 12 months. Secondary endpoints included other MDS-UPDRS subscores at 6, 12, and 14 months and doses of levodopa equivalent.
A total of 156 patients 40 to 75 years old with early Parkinson’s disease (diagnosed less than 3 years earlier) on stable symptomatic medications without motor complications were randomized 1:1 (with 78 assigned to each group) to subcutaneous injections of 20 µg lixisenatide or placebo once daily for 12 months, followed by a 2-month washout period. Patients randomized to lixisenatide received 10 μg/day for 14 days and then 20 μg/day administered by once-daily subcutaneous injections for 12 months. If patients were unable to tolerate the dose of 20 μg/day, it would have been reduced to 10 μg/day. Patients were expected to remain on a stable dosage of antiparkinsonian medications for at least the first 6 months of the trial, and optimally for the entire 14 months of follow-up. An interesting feature of the trial was that the drug was tested both during ON and OFF levodopamine periods.
MDS-UPDRS scores at baseline were approximately 15 in both groups.
At 12 months, scores on the MDS-UPDRS had changed by −0.04 points (indicating improvement) in the lixisenatide group and 3.04 points (indicating worsening disability) in the placebo group.
At 14 months, after a 2-month washout period, the mean MDS-UPDRS motor scores in the off-medication state were 17.7 (95% CI, 15.7 to 19.7) with lixisenatide and 20.6 (95% CI, 18.5 to 22.8) with placebo. Other results relative to the secondary endpoints did not differ substantially between the groups. Nausea occurred in 46% of participants receiving lixisenatide, and vomiting occurred in 13%.
What is lixisenatide?
Lixisenatide is a once-daily injectable GLP-1R agonist that is used in the treatment of Type 2 diabetes (brand name ‘Lyxumia’ in the EU and ‘Adlyxin’ in the USA). Lixisenatide can cross the blood-brain barrier.
What is a GLP-1R agonist?
Glucagon-like peptide 1 receptor agonists (or GLP-1R agonists) are a class of drugs used in the treatment of Type 2 diabetes. They act by mimicking the action of a naturally produced gut hormone called GLP-1 in the body. GLP-1 is produced by cells lining the intestines when food and drink is consumed, stimulating insulin to be released by the pancreas. Insulin helps cells absorb glucose from our food (sugar) to be used as energy.
The LixiPark study was led by Prof Olivier Rascol (University of Toulouse) and Prof Wassilios Meissner (University of Bordeaux). LixiPark is a multicenter trial performed at 21 centers of the French NS-Park/FCRIN network.
The study, sponsored by the Toulouse University Hospital, was co-funded by UK charity Cure Parkinson’s, with Van Andel Institute (VAI; in Michigan, US), and the French Ministry of Health, with drug and placebo support from pharmaceutical company Sanofi.
The LixiPark trial was a phase 2 clinical trial. Phase 2 studies often include measures of efficacy to get an idea of whether a treatment is doing what it is supposed to achieve. Phase 3 is the last stage of clinical testing and involves a very large cohort of people affected by the disease being tested for a long time to determine the effectiveness of the potential treatment alongside its long-term safety. A Phase III clinical trial of Lixisenatide in Parkinson's is ongoing.
What will happen next?
UK's Cure Parkinson’s is working with the investigators to plan the next phase of development of GLP-1R agonists for Parkinson’s. They are also awaiting the results of the phase 2 Stockholm study and the phase 3 UK trial of exenatide, another GLP-1R agonist, in Parkinson’s. These results are expected later this year and they will help to inform the next steps for this class of drugs.
Can Parkinson’s patients take lixisenatide?
Lixisenatide is still considered to be experimental for use in Parkinson’s and more research is required. There are currently no GLP-1R agonists, including lixisenatide, that are approved for use in Parkinson’s. GLP-1R agonists are also currently considered an experimental class of drugs for Parkinson’s.
There is a wide range of subtle differences between the broader class of GLP-1R agonists and they have not all been tested in Parkinson’s. Some GLP-1R agonists significantly reduce body weight (which might add concerns of frailty for people with Parkinson’s). Some GLP-1R agonists do not cross the blood-brain barrier very well and therefore are not able to have an effect in the brains of people with Parkinson’s. It is important to note that more research is required to better understand these differences in the context of a potential treatment for Parkinson’s.
When are the results expected of the Phase III exenatide trial?
The study finishes in the first half of 2024, so it is hoped that the results will be available in the second half of 2024. Researchers are also waiting for the results of a large clinical trial examining the effects of a two-year course of exenatide in people with Parkinson’s disease.
“There is a number called the 'clinically meaningful threshold' and we should appreciate that the results 'fell short' of this important metric so it is not ready for prime time in patients. In my view we should not rush to prescribe this drug or to try to creatively acquire it for our patients. We have been down this road many times including leukemia drugs, cough syrups and lithium for Parkinson. The data is not yet there to proceed to prescribing,” Michael S. Okun, MD, national medical advisor for the Parkinson's Foundation, told NeurologyLive®. “More importantly, the weight loss associated with GLP-1’s is not desirable in the majority of cases of Parkinson disease and the nausea and vomiting will not be a welcome symptom. The drug and trial is a step in the right direction, though there is much work to do.”
Will effects last?
David Standaert, a neurologist at the University of Alabama at Birmingham, who was not involved in the trial, says it’s important to know whether the effect will last beyond a year.
“We’re all cautious. There’s a long history of trying different things in Parkinson’s that ultimately didn’t work,” he says. A difference of three points in the rating score is a small change, one that many people with Parkinson’s would struggle to notice. What happens at 5 years? Is it 15 points then, or is it still 3? If it’s still 3, then this is not worth it.” He said.
More questions
A bit astonishing is that apparently, French researchers did not record any imaging biomarkers in the study to monitor disease progression and changes with drug administration. Since the trial was conducted in France, data collection regarding race or ethnic group is prohibited by law. The authors only tested 1 dose of lixisenatide, and thus other doses might have better or worse effects in patients with Parkinson’s disease.
As GLP-1 is a naturally produced hormone in the gut, it would have been interesting to search for changes in microbiome. It is well known since Heiko Braak that alpha-synuclein propagation starts in the gut.
GLP-1 inhibitors are also known to reduce inflammation, was inflammation measured? It is not reported.
Among the authors, there were many consultants, who had worked for several pharmaceutical companies, including Abbvie, that has an interest in drugs for Parkinson's disease.
 Bien qu'il y est eu de nombreuses études sur la sujet, des chercheurs Lithuaniens ont cherché à évaluer la prévalence des hallucinations mineures dans une cohorte composée de patients atteints de maladie de Parkinson et de témoins sains.
Bien qu'il y est eu de nombreuses études sur la sujet, des chercheurs Lithuaniens ont cherché à évaluer la prévalence des hallucinations mineures dans une cohorte composée de patients atteints de maladie de Parkinson et de témoins sains.
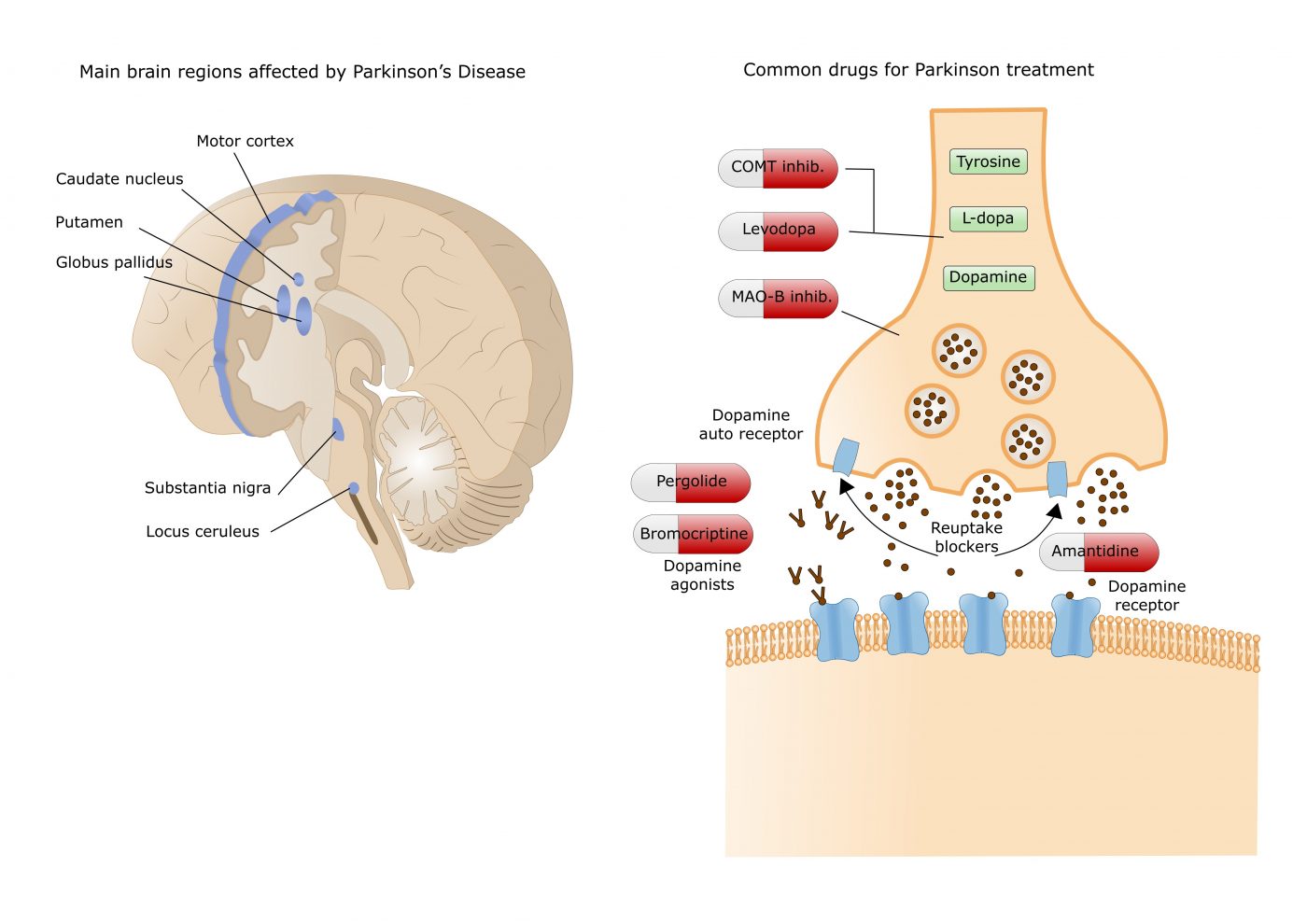 Currently available dopamine treatments work either by increasing dopamine levels (e.g. levodopa, COMT and MAO-B inhibitors) or by directly activating dopamine receptors in the striatum, an area deep in the brain.
Currently available dopamine treatments work either by increasing dopamine levels (e.g. levodopa, COMT and MAO-B inhibitors) or by directly activating dopamine receptors in the striatum, an area deep in the brain. The results of this recent trial add further evidence to exenatide’s potential as a drug that might slow the progression of Parkinson’s and pave the way for the larger phase III clinical trial that is currently underway;
The results of this recent trial add further evidence to exenatide’s potential as a drug that might slow the progression of Parkinson’s and pave the way for the larger phase III clinical trial that is currently underway; Bladder issues are unfortunately common in ALS patients. These issues arise because ALS affects the nerve cells that control the muscles in bladder and sphincter.
Bladder issues are unfortunately common in ALS patients. These issues arise because ALS affects the nerve cells that control the muscles in bladder and sphincter. De même il existe une relation entre le métabolisme des glucides et celui du fer. Ce qui suggère qu'un métabolisme anormal des glucides puisse avoir une relation avec l'apparition de la maladie de Parkinson.
De même il existe une relation entre le métabolisme des glucides et celui du fer. Ce qui suggère qu'un métabolisme anormal des glucides puisse avoir une relation avec l'apparition de la maladie de Parkinson. The study shows systematic differences and potential directional biases between research and real-world datasets. Patients in research populations are diagnosed much earlier, start levodopa and other Parkinson's medications earlier, and show slower changes in clinical scales of motor and cognitive progression. Real-world-based populations are diagnosed at older ages, start medications later than research cohorts, and experience more rapid changes in clinical scales.
The study shows systematic differences and potential directional biases between research and real-world datasets. Patients in research populations are diagnosed much earlier, start levodopa and other Parkinson's medications earlier, and show slower changes in clinical scales of motor and cognitive progression. Real-world-based populations are diagnosed at older ages, start medications later than research cohorts, and experience more rapid changes in clinical scales.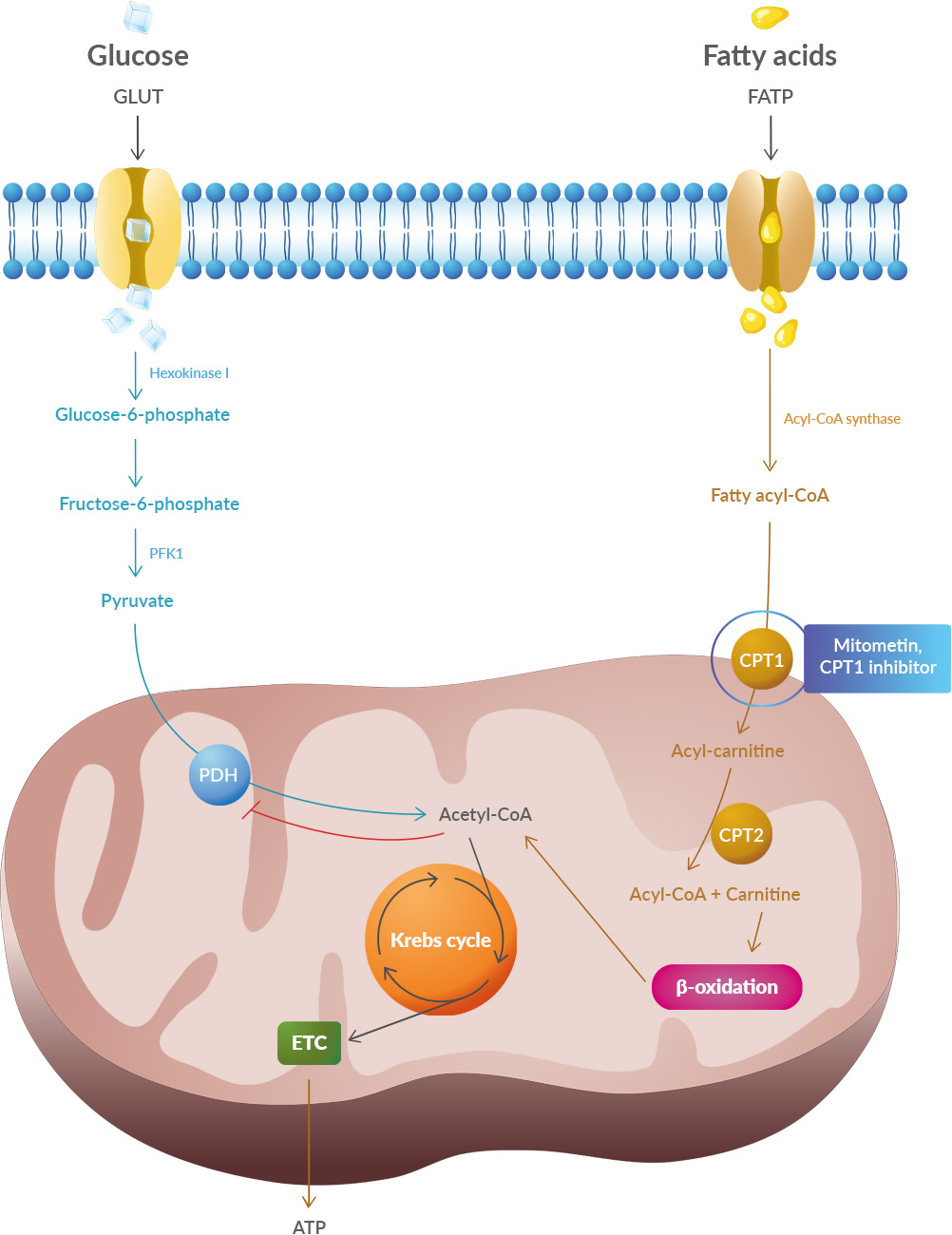 Studies have shown that oxidative stress and endoplasmic reticulum stress are correlated and can lead to protein misfolding (Abramov et al., 2020). Accumulation of misfolded proteins causes cellular damage and mitochondrial dysfunction and is associated with a range of neurodegenerative diseases, including ALS (misfolded SOD1, TDP-43, C9orf72) (McAlary et al., 2020), Parkinson's disease (misfolded α-synuclein) and Alzheimer disease (misfolded Aβ and Tau) (Abramov et al., 2020).
Studies have shown that oxidative stress and endoplasmic reticulum stress are correlated and can lead to protein misfolding (Abramov et al., 2020). Accumulation of misfolded proteins causes cellular damage and mitochondrial dysfunction and is associated with a range of neurodegenerative diseases, including ALS (misfolded SOD1, TDP-43, C9orf72) (McAlary et al., 2020), Parkinson's disease (misfolded α-synuclein) and Alzheimer disease (misfolded Aβ and Tau) (Abramov et al., 2020).