The anticipation of progression of Alzheimer's disease is crucial for evaluations of secondary prevention measures thought to modify the disease trajectory. However, it is difficult to forecast the natural progression of Alzheimer's disease, notably because several functions decline at different ages and different rates in different patients.
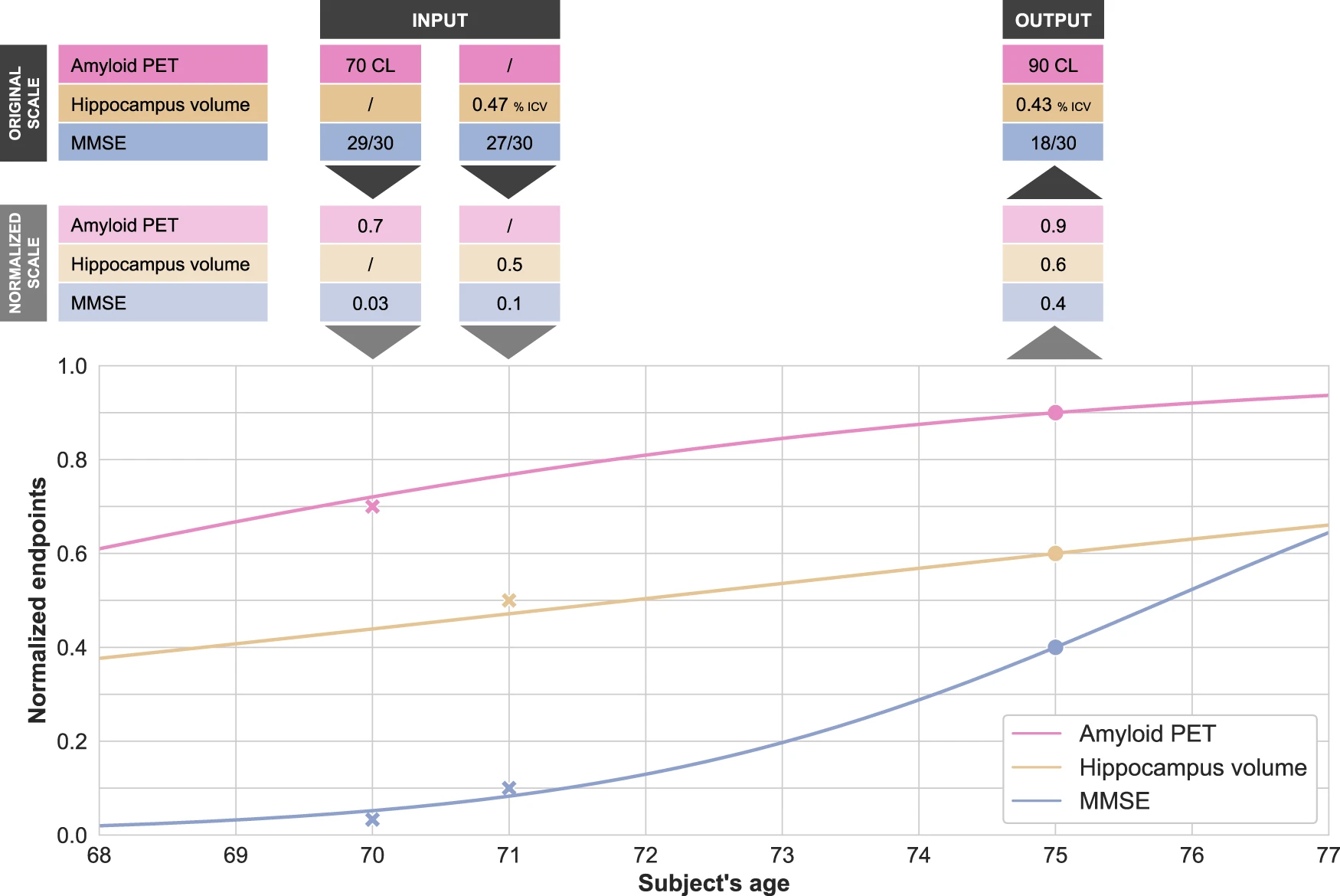 Disease modeling uses computational and statistical methods to address this question. These models learn the variability of disease progression from observational longitudinal cohort data and can then predict the progression of patients from their historical data. They require various clinical or biomarker assessments at one or several time points as input.
Disease modeling uses computational and statistical methods to address this question. These models learn the variability of disease progression from observational longitudinal cohort data and can then predict the progression of patients from their historical data. They require various clinical or biomarker assessments at one or several time points as input.
European authors evaluate in Nature communications Alzheimer's disease Course Map, a statistical model predicting the progression of neuropsychological assessments and imaging biomarkers for a patient from current medical and radiological data at early disease stages.
Alzheimer's disease Course Map is a spatiotemporal atlas of Alzheimer's disease progression. It summarizes the variability in the progression of a series of neuropsychological assessments, the propagation of hypometabolism and cortical thinning across brain regions and the deformation of the shape of the hippocampus. The analysis of these variations highlights strong genetic determinants for the progression, like possible compensatory mechanisms at play during disease progression. AD Course Map also predicts the patient's cognitive decline with a better accuracy than the 56 methods benchmarked in the open challenge TADPOLE. Finally, AD Course Map is used to simulate cohorts of virtual patients developing Alzheimer's disease.
The scientists from France, Germany and Italy propose a software tool using a disease progression model for participant selection in clinical trials. The goal is to enrich the selected population of participants likely to display progression during the trial, a concept called prognostic enrichment.
The scientists tested the method on more than 96,000 cases, with a pool of more than 4,600 patients from four continents.
They measured the accuracy of the method for selecting participants displaying a progression of clinical endpoints during a hypothetical trial.
They show that enriching the population with the predicted progressors decreases the required sample size by 38% to 50%, depending on trial duration, outcome, and targeted disease stage, from asymptomatic individuals at risk of Alzheimer's disease to subjects with early and mild Alzheimer's disease.
In conclusion this method introduces no biases regarding sex or geographic locations and is robust to missing data. It performs best at the earliest stages of disease and is therefore highly suitable for use in prevention trials.
