Several ongoing efforts are to design and test drugs targeting cellular stress in ALS. The reasoning is that in ALS there is an abnormally prolonged cellular stress response that nearly stops the cell activity (because of lack of nutrients, including because of insulin resistance) and therefore creates a kind of traffic jam at the entrance of the Endoplasmic Reticulum. This would nicely explain why unfolded proteins are found in the cytoplasm of Central Nervous System cells.
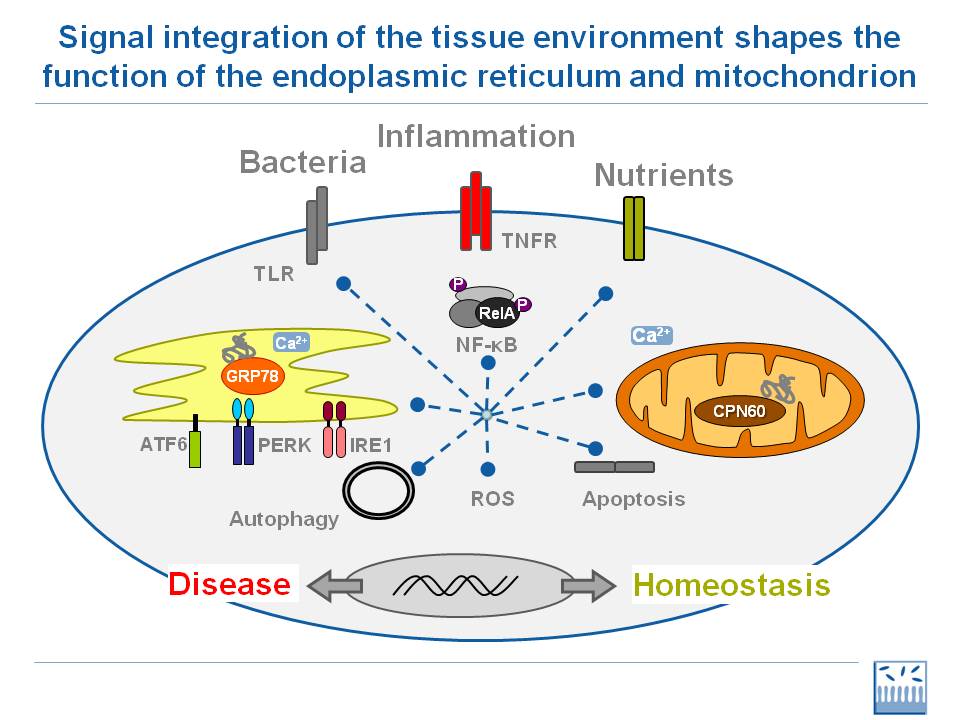 Yet it's been known for a long time that granules are formed when cells encounter a stress event. Stress granules disappear as soon as the cell resumes its normal functioning.
Yet it's been known for a long time that granules are formed when cells encounter a stress event. Stress granules disappear as soon as the cell resumes its normal functioning.
Scientists in Japan wanted to know if TDP-43 unfolded, mislocalized aggregations have some relation with stress granules. TDP-43 is an RNA-binding protein with various functions in RNA metabolism.
Previous studies have proposed a connection between TDP-43 inclusions and stress granules as often they colocalize.
This study investigated this relationship by examining the presence of a stress granule marker protein called HuR in ALS patients' anterior horn cells (AHCs). It included cases of sporadic ALS with different disease durations, comparing them to normal controls. The results showed a decrease in HuR immunoreactivity in AHCs of ALS patients, especially in standard-duration cases.
Furthermore, the study found that TDP-43 inclusions appear to have their origins in structures resembling stress granules, especially during the early stages of inclusion formation. These structures, termed DPCS (dot-like punctate cytoplasmic structures), were found to be HuR-positive and associated with the ribosome-like structures near the endoplasmic reticulum.
In conclusion, this research suggests that TDP-43 aggregation in ALS may begin with the involvement of stress granules. Understanding these early events may have implications for future ALS therapies.
While this research is not making huge claims (contrary to most low-quality scientific publications) it will certainly help to fund drug development for helping cells recover from abnormal cellular stress response.
