Cells that are damaged or dysfunctional and pose a threat are either eliminated by cell death or undergo a generally irreversible state called senescence. Senescent cells usually do not divide, but they can persist in tissues and contribute to aging and cancer. Senescent cells often become larger and may have higher energy requirements than healthy cells.
Chemotherapy that damages the DNA of cancer cells can lead cells to death or senescence. Cellular entry into senescence benefits an organism by inhibiting the development of cancer by preventing the division of cells that have accumulated significant DNA damage.
Tonnessen-Murray and colleagues investigated the effects of chemotherapy-induced senescence in breast cancer cells in mice treated with doxorubicin, a chemotherapeutic drug.
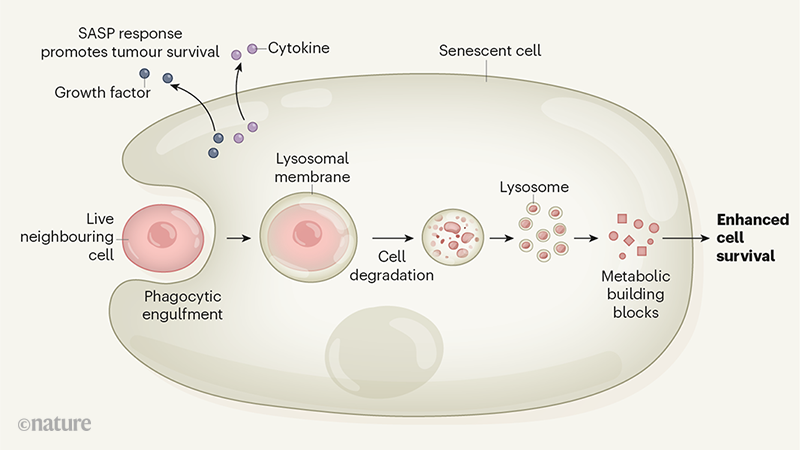
These senescent cells absorb and digest neighboring living cells. The cells are engulfed by a process that has the molecular characteristics of phagocytosis, a process used by immune cells. Once ingested, the cells are enveloped in the membrane of an organelle called lysosome and digested.
This degradation provides metabolic building blocks for the cell. Senescent cells that have ingested their neighbors survive longer than senescent cells that have not. This suggests that the metabolic building blocks recovered from lysosomal digestion of neighboring cells were used by senescent cells to promote their survival.
This surprising discovery highlights the complexity of the regulation of cell death in multicellular animals. Many mechanisms of cell death occur in animal tissues. These include forms of cell suicide, such as apoptosis, which leads to fragmentation of individual cells, and regulated forms of necrotic cell death that cause cell disruption. Some cases of engulfing cells occur through at least two distinct mechanisms:
- One is a form of cell suicide called entosis, in which cells invade a neighboring cell and are phagocytized by it.
- The other mechanism is cellular cannibalism resembling the process used by immune system cells such as macrophages to ingest and destroy dying cells.
The authors analyzed the gene expression profile of cancer cells treated with chemotherapy drugs (most of these cells were senescent) and found that the characteristic genes for phagocytosis were expressed. This gene expression culminated in a time that correlated with cell engulfment. They also observed that senescent cells engulfed dead cells added in vitro, providing a further clue that senescent cells engulf cells by phagocytosis.
Cellular cannibalism in cancers has already been reported. However, Tonnessen-Murray and his colleagues specifically identified an association between cannibalism and senescence, and they show that this phenomenon could make a substantial contribution to the persistence of senescent cells in cancerous tissues. Their results suggest that cellular cannibalism may be an activity that is largely associated with the induction of senescence, rather than being related to particular types of cancer or the status of proteins such as p53. It will be important to study if cannibalism is related to senescence in other contexts, for example the proteopathies that are found in neurodegenerative diseases, which moreover was suggested more than 10 years ago by Todd E Golde, and Victor M Miller. Since 2018, it has been known that senescent astrocytes and microglia accumulate in the brain in the context of diseases associated with Tau protein.

