Post Intensive Care Syndrome (PICS) describes a set of disorders that are common in patients with severe illness and / or intensive care. Since the majority of the literature in intensive care medicine focuses on short-term outcomes (for example, survival), understanding of the patient's long-term development is relatively limited, since the latter is then considered to be healed.
Cognitive impairment includes deficits in memory, attention, speed of mental processing and problem solving. These impairments affect up to 80% of people who have experienced a serious illness. Most patients' symptoms improve or even disappear completely within the first year after treatment in the intensive care unit.
The underlying pathophysiology of cognitive impairment in critical care survivors is not well understood, but prolonged inflammation can play an important role
High Mobility Group Box 1 (HMGB1), a protein released in tissue damage and during severe inflammation, has been shown in laboratory animals to remain in high concentration long after the trauma and can cause inflammation hippocampal and cognitive impairment.
 Source: Life Science Databases(LSDB) via Wikipedia.
Source: Life Science Databases(LSDB) via Wikipedia.
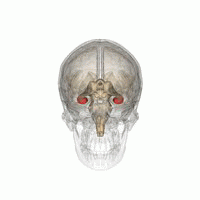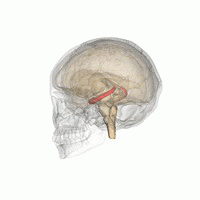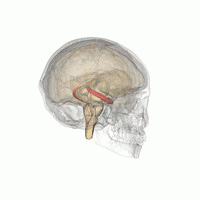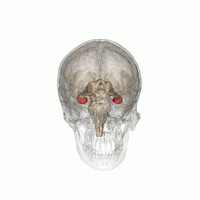
Humans and other mammals have two seahorses, one on each side of the brain. The hippocampus is part of the limbic system. In Alzheimer's disease, the hippocampus is one of the first areas of the brain to be damaged; short-term memory loss and disorientation are among the first symptoms. People with extensive bilateral hippocampal lesions may suffer from anterograde amnesia: the inability to form and retain new memories.
The form of neural plasticity known as long-term potentiation (LTP) was originally discovered in the hippocampus and has often been studied in this structure. LTP is considered to be one of the main neural mechanisms by which memories are stored in the brain.
Anti-HMGB1 treatment given several days after a serious illness can reduce cognitive decline in mice
Researchers at the Karolinska Institutet in Sweden conducted a 6-month prospective follow-up study of HMGB1 plasma levels and cognitive function in survivors of intensive care (clinical trial NCT02914756). 917 patients admitted to intensive care were screened, of which 100 patients were included in the clinical trial, and they were subjected to cognitive function tests and to the measurement of plasma levels of HMGB1 at 3 and 6 months after discharge
The observations were made in these patients show a significant elevation of HMGB1 plasma at 3 and 6 months after discharge, and is associated with cognitive dysfunction.
The cellular source of this systemic HMGB1 is unknown, but it should be noted that HMGB1 is usually secreted by immune cells (such as macrophages, monocytes and dendritic cells) as a mediator of the cytokines of inflammation.
Given these well-established pro-inflammatory properties of extracellular HMGB1, this suggests continued inflammation without resolution of the inflammation. In light of the experimental results on the attenuation of cognitive dysfunction in laboratory animals by anti-HMGB1 therapy, it is tempting to ask whether blocking the pro-inflammatory activity of HMGB1 in ICU survivors could improve cognitive outcomes.
